alvarez/E+ via Getty Images
A Quick Take On Mineralys Therapeutics
Mineralys Therapeutics (MLYS) has filed to raise $119 million in an IPO of its common stock, according to an S-1 registration statement.
The firm is a clinical-stage biopharma developing treatments for diseases due to abnormally elevated aldosterone.
MLYS has produced positive Phase 2 efficacy results for its primary candidate.
I’ll provide an update when we learn more IPO details from management.
Mineralys Overview
Radnor, Pennsylvania-based Mineralys Therapeutics was founded to develop lorundrostat, an aldosterone inhibitor that promises to reduce uncontrolled hypertension [uHTN] in patients unable to achieve blood pressure below 130/80.
Management is headed by Chief Executive Officer Jon Congleton, who has been with the firm since November 2020 and was previously CEO of Impel NeuroPharma and, before that, CEO of Nivalis Therapeutics.
The firm’s lead candidate, lorundrostat, has produced positive results in a Phase 2 efficacy trial that evaluated 200 subjects.
The company plans another Phase 2 trial in 2023 with the candidate ‘used as an add-on therapy to background treatment of two or three antihypertensive medications in up to approximately 240 adults.’
Below is the current status of the company’s drug development pipeline:
Company Pipeline (SEC)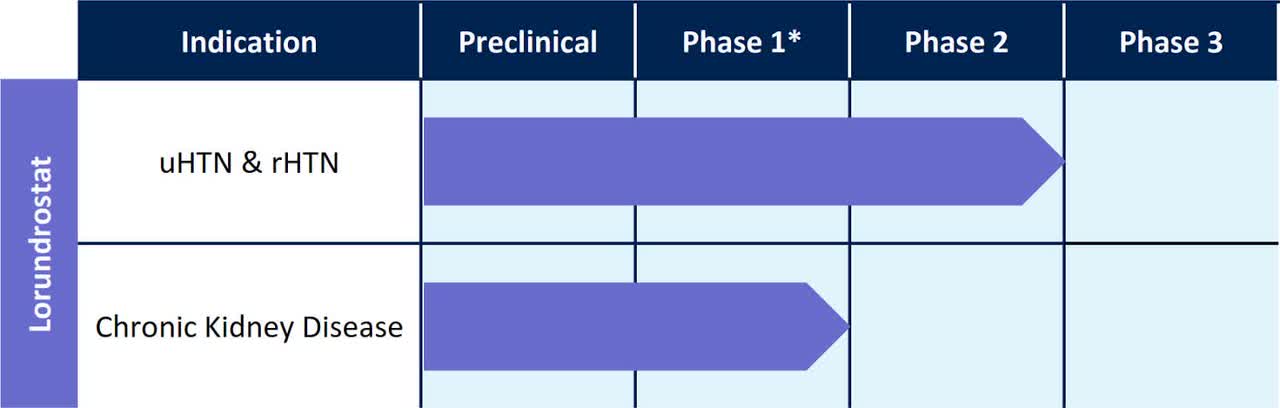
Mineralys has booked fair market value investment of $158.8 million as of September 30, 2022 from investors including Catalys Pacific Fund, Samsara BioCapital, HBM Healthcare, Adams Street Partners, BioDiscovery and RA Capital Management.
Mineralys’ Market & Competition
According to a 2019 market research report by BCC Research, the global market for antihypertensive drugs was an estimated $26.3 billion in 2018 and is forecast to reach $27.8 billion by the end of 2023.
This represents a forecast CAGR (Compound Annual Growth Rate) of 1.1% from 2018 to 2023.
Key elements driving this expected growth are an aging global population which will lead to a greater incidence of hypertension and the resulting demand for improved treatments.
Also, according to iHealthcareAnalyst, between 2000 and 2010, the prevalence of hypertension in high-income countries was reduced by 2.6%, while it increased by 7.7% in low- and middle-income countries.
This was due to continuing ‘obstacles in healthcare delivery and limited resources’ in less-developed countries.
Major competitive vendors that provide or are developing related treatments include:
-
Boehringer Ingelheim
-
CinCo
-
Damian Pharma
-
PhaseBio (OTC:PHASQ)
-
Idorsia (OTCPK:IDRSF)
-
Quantum Genomics (OTCPK:QNNTF)
-
Ionis (IONS)
-
Alnylam (ALNY)
-
Sihuan Pharmaceutical Holdings (OTCPK:SHPHF)
-
KBP BioSciences
-
AstraZeneca (AZN)
-
Johnson & Johnson (JNJ)
-
Merck (MRK)
-
Novartis (NVS)
-
Pfizer (PFE)
-
Takeda Pharmaceuticals (TAK)
Mineralys Therapeutics Financial Status
The firm’s recent financial results are typical of a clinical-stage biopharma in that they feature no revenue and significant R&D and G&A expenses associated with its drug development efforts.
Below are the company’s recent financial results:
Statement Of Operations (SEC)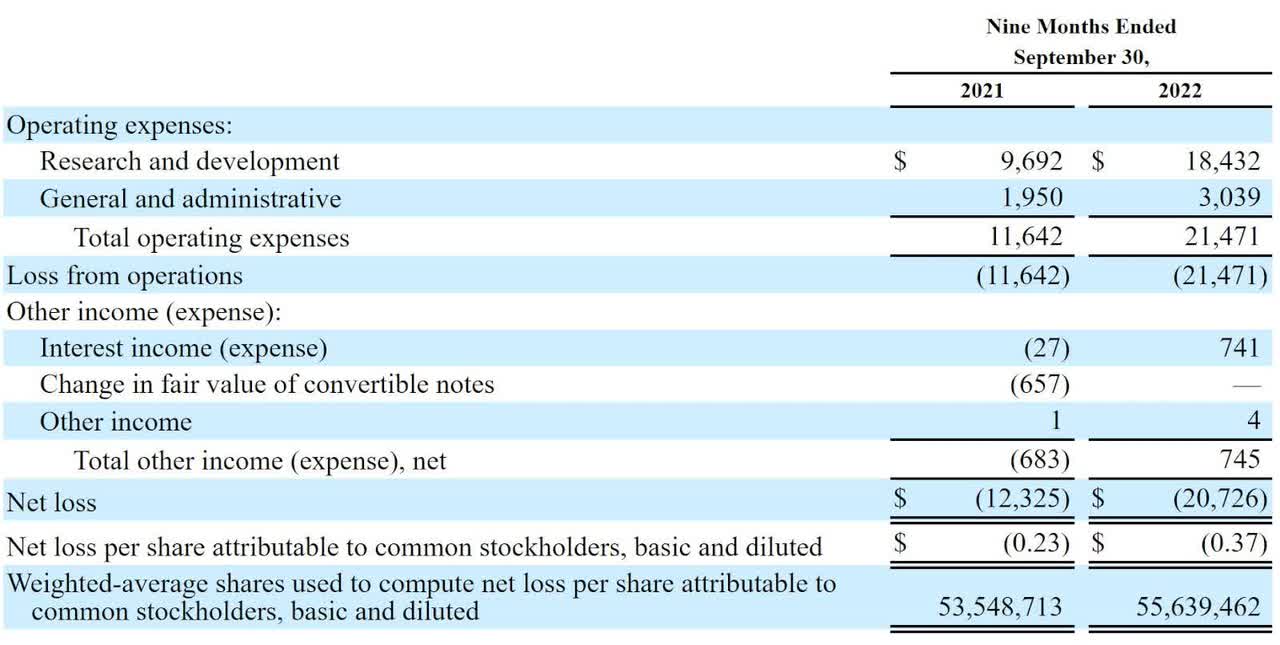
As of September 30, 2022, the company had $49 million in cash and $7.2 million in total liabilities.
Mineralys Therapeutics IPO Details
Mineralys intends to raise $119 million in gross proceeds from an IPO of its common stock, although the final figure may vary.
No existing shareholders have indicated an interest in purchasing shares at the IPO price, although this element may become a feature of the IPO if disclosed in a future filing.
Management says it will use the net proceeds from the IPO as follows:
to fund the research and development of lorundrostat, and the remainder for working capital and general corporate purposes.
(Source – SEC)
Management’s presentation of the company roadshow is not available.
Regarding outstanding legal proceedings, management says the firm is not currently a party to any legal proceeding that it calls ‘material.’
Listed bookrunners of the IPO are BofA Securities, Evercore ISI and other investment banks.
Commentary About Mineralys’ IPO
MLYS is seeking U.S. public capital market investment to fund the advancement of its pipeline of drug treatment programs.
The firm’s lead candidate, lorundrostat, has produced positive results in a Phase 2 efficacy trial that evaluated 200 subjects.
The company plans another Phase 2 trial in conjunction with other existing treatments.
Additionally, in the future, management believes it can develop lorundrostat for the treatment of chronic kidney disease. Chronic kidney disease treatment is expected to reach $79 billion in annual value by 2026.
The market opportunity for treating hypertension is quite large and expected to grow at a relatively low rate of growth in the coming years.
Management has not disclosed any major pharma firm collaboration relationship.
The company’s investor syndicate includes a number of well-known institutional venture capital and private equity firms active in the life science sector.
BofA Securities is the lead underwriter and IPOs led by the firm over the last 12-month period have generated an average return of 80.5% since their IPO. This is a top-tier performance for all major underwriters during the period.
When we learn more information about the IPO’s pricing and valuation assumptions, I’ll provide a final opinion.
Expected IPO Pricing Date: To be announced.


Be the first to comment